- Blog
- Euro cccam test
- Best 5e character builder app
- Sims 3 kinky world animal animations
- Mp3 download
- Singstar songs download
- Critical ops mod money
- Quickverse Bible Software For Windows 10
- Monster musume hentai uncensored
- Minecraftr gulliver mod 1-7-10
- Eviews 10 patch
- Ns2 np3
- Spyder 4 elite led monitor

#Ns2 np3 pdf#
MCQ on Periodic Classification of Elements class 11 PDF with Answersġ.
#Ns2 np3 download#
We have provided these questions in PDF format which you can Download by clicking the link as “classification of elements and periodicity in properties mcq pdf” provide below. These MCQs are very helpful in your preparation for the National Eligibility cum Entrance Test-UG (NEET) and JEE mains. So these classification of elements and periodicity in properties neet questions cover all the topics of Classification of Elements and Periodicity in Properties. Preparation of these questions has been taken from various books including NCERT, Previous year’s question papers, and model papers. These questions are based on the chapter of Classification of Elements and Periodicity in Properties.

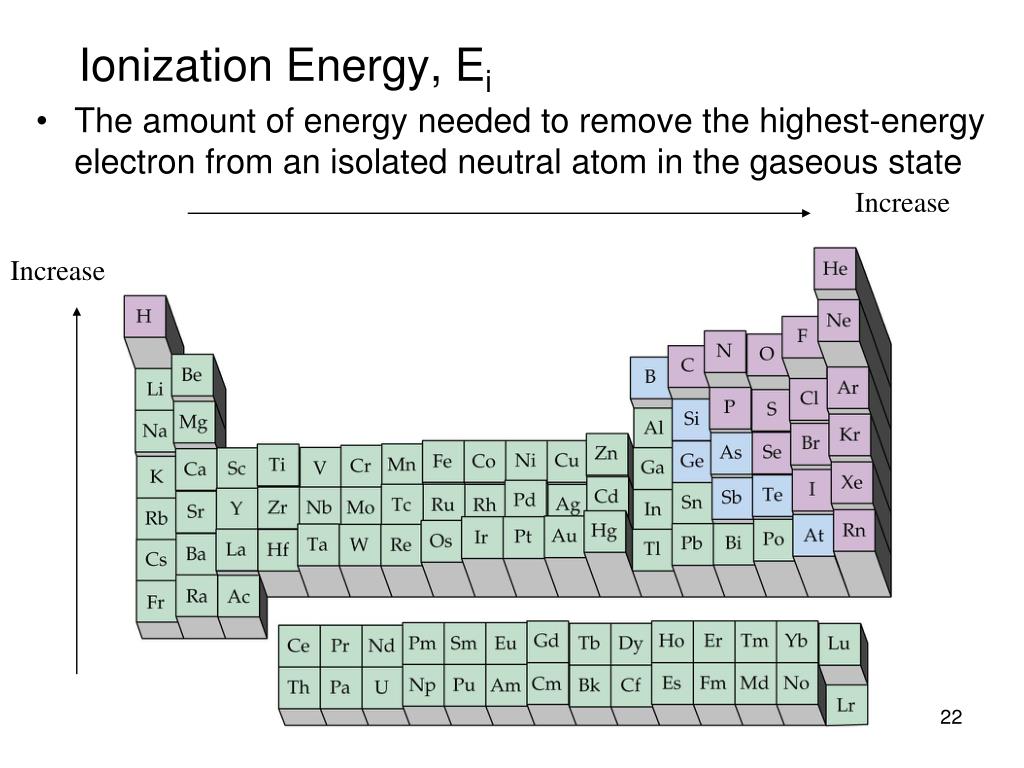
Physical properties incorporate physical state, boiling and melting points, metallic character, allotropy, and density.These MCQ on Periodic Classification of Elements Class 11 pdf with Answers are prepared by our LiveMCQs team for NEET. The electro negativity diminishes bit by bit on moving down the group because of the increase in atomic radius. This is because of the progressive increment in the nuclear size.Įlectro negativity is the inclination of a particle to pull in a shared pair of electrons more towards itself. As we move down the group, the ionization enthalpy values diminish. This is because of their higher atomic charge, smaller nuclear radii, and stable half-filled electronic setups. These elements demonstrate higher values of ionization enthalpy when contrasted with group 14 elements. Moving down the group, the ionic radii, and atomic radii increases because of the expansion of another main energy level in each progressive element. 3: Electronic configuration of group 15 elements Because of the precisely half-filled electronic arrangement of the 'n p' subshell, the elements of this group are genuinely steady and stable.Įxample: Dinitrogen is an inactive gas under ordinary conditions.įig. There are five electrons in the valence shell of these elements. Thus, the valence shell electronic arrangement for these elements becomes ns2 np3. The separating electron in the elements of group 15 enters the n p subshell. The rest of the elements of the group, that is, arsenic, antimony, and bismuth, mostly happen as sulfides.Įxample: Stibnite, Arsenopyrite, and bismuth glance. Phosphoproteins are available in egg yolk, milk, and bone marrow. Around 60% of bones and teeth are made out of phosphates. Phosphate groups are constituents of nucleic acids, that is, DNA and RNA. Phosphorus is a fundamental constituent of animal and plant matter. Example: Fluoroapatite, Chlorapatite, and Hydroxyapatite are a few. In the consolidated state, it occurs as minerals as phosphates.
It is the eleventh most copious element in the crust of the earth. The following element in the group is phosphorus. It is additionally found as the fundamental constituent of proteins, nucleic acids, amino acids, and catalysts. Minerals of Nitrogen: Indian saltpeter and Chile saltpeter.
#Ns2 np3 free#
It is the primary member of this group and happens in a free state as a diatomic gas, N2. Nitrogen is the real constituent of the world's air, and records for 78% of it by volume. Group 15 elements include nitrogen, phosphorus, arsenic, antimony and bismuth. 1: Elements of group 15 with their atomic number, electronic configuration, group number and period number.

P-block elements are generally non-metals, while the remaining are metalloids and metals.įig. Disregarding the way that helium does not have p orbitals, it is a p-block element since it takes after that of other p-block elements of the eighteenth group concerning their physical and compound properties. The general valence shell electronic setup of group fifteen elements is ns 2, np 1-6. In this manner, in these elements, the n p subshell is step by step filled. In the iotas of p-block elements, the separating electron enters the valence p subshell. The p-block elements are put to the right-hand side of the periodic table in groups from 13 to 18.
